Just nine months post LumineticsCore® expansion, OSF Healthcare identified diabetic retinopathy in 25% of nearly 1,500 patients.
In April 2018, Digital Diagnostics became the first company to receive FDA clearance for an AI diagnostic platform that makes a diagnosis without physician input at the point-of-care. Since then, Digital Diagnostics has diligently worked from within the healthcare system to establish autonomous diagnosis as the standard of care, as supported by the American Diabetes Association.1
The continued hard work to ensure the healthcare ecosystem understands and supports commercial adoption of Digital Diagnostics’ flagship product, LumineticsCore® (formerly IDx-DR) is paramount to meet the unmet need of eye exams for diabetes at primary care. The AI diagnostic system autonomously diagnoses patients for diabetic retinopathy, is in use at over 70 health systems, including OSF Healthcare, a not-for-profit Catholic healthcare organization.
The partnership between Digital Diagnostics and OSF Healthcare has been the subject of several articles, blogs, videos, and podcasts, including New AI Technology Helps Central Illinois Diabetics. Published on the Fox Illinois website, the article highlights patients’ positive reactions to LumineticsCore® in OSF doctors’ offices.
“But had OSF not had that [LumineticsCore® exam], I would have put that off a lot longer, like it’s there, you know people should do it,” said Huffman, an OSF patient.
The partnership between Digital Diagnostics and OSF Healthcare also made headlines, when Vermillion County First published A New Vision for Diabetes-Related Eye Exams Coming to OSF in Champaign. In the article, Dr. Mark Meeker, DO, FACP, CPE, Vice President & Chief Medical Officer at OSF HealthCare, discussed the partnership between Digital Diagnostics and OSF Healthcare and how LumineticsCore® is changing the way diabetic retinopathy is diagnosed.
“We know from the pilot we launched through OSF Innovation at eight locations last May, that nearly one-quarter of all patients with diabetes who received the [LumineticsCore®] exam tested positive for diabetic retinopathy,” said Dr. Meeker. He further added, “There are interventions such as laser treatment and medication that can make a difference if an individual tests positive early enough, but prevention through better diabetes management is our goal.”
The Illinois Business Journal highlighted the flourishing partnership between Digital Diagnostics and OSF Healthcare, calling attention to the fact that OSF Healthcare utilizes advanced technology for people with diabetes to get an exam to check for diabetic retinopathy when visiting their primary care provider. In the article, Dr. Meeker again explained the benefits of LumineticsCore®.
“Early detection and prevention can prolong and even save vision,” Dr. Meeker stressed.
OSF Healthcare also released an episode of its podcast, Health Accelerated, during which, Dr. Meeker discussed in detail how OSF HealthCare is using Digital Diagnostics’ flagship product, LumineticsCore®, to detect diabetic retinopathy, ultimately saving patients’ sight, and, perhaps, their health.
“It’s really a win-win-win all the way around,” said Dr. Meeker.
In May 2024, OSF Healthcare released a blog entitled “Quick, Easy Eye Exam Helps Prevent Diabetic Retinopathy.” Throughout the blog, Dr. Meeker stressed the importance of making eye exams for diabetes as convenient as possible. He went on to explain how LumineticsCore® makes in-office testing quick and easy, all while providing more accurate results than typical eye exams given in a primary care setting.
“The AI can see the early changes of diabetic retinopathy that we may not be able to see with our own eyes,” Dr. Meeker said. He continued, “There’s nothing as good as prevention. And the way you prevent it is to tightly control the diabetes to begin with. So as soon as we see those retinopathy changes, if we can really get the attention of the patient to really pay attention to their sugar control, we can decrease that progression through prevention, not just through treatment.”
Another OSF Healthcare blog, “AI Platform Provides Easy Access to Eye Disease Diagnosis for OSF Patients with Diabetes,” reports significant progress at OSF Healthcare: Following successful integration at eight initial sites, OSF Healthcare expanded the use of LumineticsCore® to 24 additional sites. Just nine months post-expansion, OSF Healthcare identified diabetic retinopathy in 25% of nearly 1,500 patients.
“We are always seeking new ways to innovate and improve access to cutting-edge diagnostics for our patients so we can help them better manage their health conditions, including diabetes which impacts nearly 66,000 OSF patients,” said Dr. Meeker.
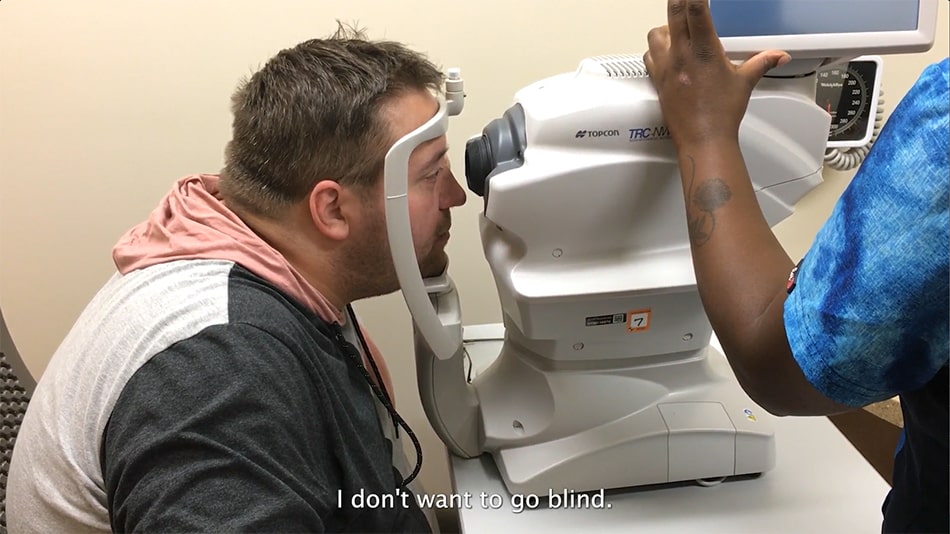
In November 2024, OSF Healthcare published “Keep an Eye on Diabetic Retinopathy,” offering a unique patient perspective on diabetic retinopathy and the annual eye exam for diabetes. The blog features the story of Andrew Hade, an OSF Healthcare patient who, like many with diabetes, initially skipped the recommended annual eye exam due to a lack of symptoms. It was only after undergoing the LumineticsCore® exam at OSF Healthcare that he was diagnosed with diabetic retinopathy.
“Now that I do know, I can take steps to prevent the worst possible result, which is, going blind,” Hade said. “I don’t want to go blind, so getting the test makes it so I might be able to avoid that. Not taking the test means that I wouldn’t know,” said Andrew Hade.
In that same blog, OSF Healthcare highlights how LumineticsCore® is revolutionizing early diagnosis of diabetic retinopathy within their organization by making the eye exam for diabetes more accessible for their patients.
“In the first six months of using [LumineticsCore®] technology in OSF HealthCare offices, 28 out of 100 people with diabetes tested positive, with about a 2% false-positive rate. That’s 28% of patients with diabetes who would have been at risk of losing their vision in the future if not for the [exam] to detect it early.”
Through strategic collaborations, the vision of Digital Diagnostics continues to expand; however, the mission remains the same:
To benefit patients by transforming the accessibility, affordability, and quality of global healthcare through the application of technology in the medical diagnosis process.
Note: Since the release of the above-mentioned publications, Digital Diagnostics rebranded its flagship product, IDx-DR, to LumineticsCore®. Any mention of the former product name, IDx-DR, in the linked content is in reference to LumineticsCore®. The Indications for Use and autonomous AI technology behind LumineticsCore® are unchanged.
Updated May 2025
References
- ElSayed, N. A., Aleppo, G., Aroda, V. R., Bannuru, R. R., Brown, F. M., Bruemmer, D., Collins, B. S., Cusi, K., Das, S. R., Gibbons, C. H., Giurini, J. M., Hilliard, M. E., Isaacs, D., Johnson, E. L., Kahan, S., Khunti, K., Kosiborod, M., Leon, J., Lyons, S. K., & Murdock, L. (2022). Introduction and Methodology: Standards of Care in Diabetes—2023. Diabetes Care, 46(Supplement_1), S1–S4. https://doi.org/10.2337/dc23-sint